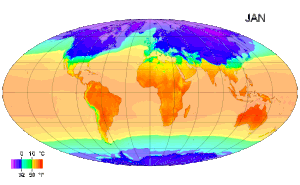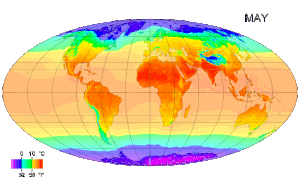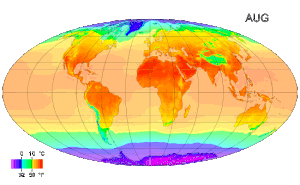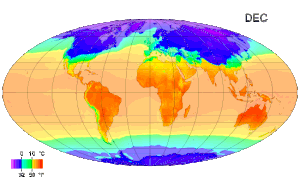
Temperature measurement - Temperature measurement using modern scientific thermometers and temperature scales goes back at least as far as the early 18th century, when Gabriel Fahrenheit adapted a thermometer (switching to mercury) and a scale both developed by Ole Christensen Rømer. Fahrenheit's scale is still in use in the United States for non-scientific applications.
Temperature is measured with thermometers that may be calibrated to a variety of temperature scales. In most of the world (except for Belize, Myanmar, Liberia and the United States), the Celsius scale is used for most temperature measuring purposes. Most scientist measures temperature using the Celsius scale and the thermodynamic temperature using the Kelvin scale, which is the Celsius scale offset so that its null point is 0K = −273.15°C, or absolute zero. Many engineering fields in the U.S., notably high-tech and US federal specifications (civil and military), also use the Kelvin and Celsius scales. Other engineering fields in the U.S. also rely upon the Rankine scale (a shifted Fahrenheit scale) when working in thermodynamic-related disciplines such as combustion.
Units
The basic unit of temperature in the International System of Units (SI) is the kelvin. It has the symbol K.
For everyday applications, it is often convenient to use the Celsius scale, in which 0°C corresponds very closely to the freezing point of water and 100°C is its boiling point at sea level. Because liquid droplets commonly exist in clouds at sub-zero temperatures, 0°C is better defined as the melting point of ice. In this scale a temperature difference of 1 degree Celsius is the same as a 1kelvin increment, but the scale is offset by the temperature at which ice melts (273.15 K).
By international agreement[25] the Kelvin and Celsius scales are defined by two fixing points: absolute zero and the triple point of Vienna Standard Mean Ocean Water, which is water specially prepared with a specified blend of hydrogen and oxygen isotopes. Absolute zero is defined as precisely 0K and −273.15°C. It is the temperature at which all classical translational motion of the particles comprising matter ceases and they are at complete rest in the classical model. Quantum-mechanically, however, zero-point motion remains and has an associated energy, the zero-point energy. Matter is in its ground state,[26] and contains no thermal energy. The triple point of water is defined as 273.16K and 0.01°C. This definition serves the following purposes: it fixes the magnitude of the kelvin as being precisely 1 part in 273.16 parts of the difference between absolute zero and the triple point of water; it establishes that one kelvin has precisely the same magnitude as one degree on the Celsius scale; and it establishes the difference between the null points of these scales as being 273.15K (0K = −273.15°C and 273.16K = 0.01°C).
In the United States, the Fahrenheit scale is widely used. On this scale the freezing point of water corresponds to 32 °F and the boiling point to 212 °F. The Rankine scale, still used in fields of chemical engineering in the U.S., is an absolute scale based on the Fahrenheit increment.